|
11/24/2023 0 Comments Charge of mole to charge of electronBurns tells Smithers to "just give him Faraday's Constant". He says "the speed of light expressed as dollars" and Mr. Burns asking Comic Book Guy how much he wants for his entire comic book inventory. The Simpsons episode " Dark Knight Court" has Mr. The faraday is not to be confused with the farad, an unrelated unit of capacitance ( 1 farad = 1 coulomb / 1 volt). ×10 4 C.Ĭonversely, the Faraday constant F equals 1 faraday per mole. One faraday of charge is the charge of one mole of elementary charges (or of negative one mole of electrons), i.e.,ġ faraday = F × 1 mol = 9.648 533 212. Its use is much less common than of the coulomb, but is sometimes used in electrochemistry. Transcribed image text: CONSIDER THIS Calculate an accurate value of Avogadros number as the ratio of the electric charge on a mole of elections (Faradays constant) divided by the charge on a single electron (see Table 2 of the Appendix Calculate the edge length of a cube containing an Avogadros number (6.02 x 10') of. Related to the Faraday constant is the "faraday", a unit of electrical charge. So we get the charge on one mole of electrons as (1.6 x 10-16) x (6.02 x 1023) 96500 C which is also called 1 Faraday. There are 6.02 x 1023 electrons per mole. 0 2 × 1 0 2 3 electrons per mole, so the charge will be ( 1. The value of F was first determined by weighing the amount of silver deposited in an electrochemical reaction in which a measured current was passed for a measured time, and using Faraday's law of electrolysis. Hi Ugendhar, Charge on a single electron is 1.6 x 10-16 Coulomb. So the faraday is basically a conversion. One can divide the amount of charge (the current integrated over time) by the Faraday constant in order to find the chemical amount of a substance (in moles) that has been electrolyzed. A faraday is the amount of charge contained by exactly one mole of electrons. One common use of the Faraday constant is in electrolysis calculations. Faradays Constant is given the symbol (F) and is defined as the charge in coulumbs (C) of 1 mole of electrons. electron charge, (symbol e ), fundamental physical constant expressing the naturally occurring unit of electric charge, equal to 1.602176634 × 10 19 coulomb. Because 1 mole contains exactly 6.022 140 76 ×10 23 entities, and 1 coulomb contains exactly C / e = 10 19 / 1.602 176 634 elementary charges, the Faraday constant is given by the quotient of these two quantities:į = N A / 1/ e = 9.648 533 212 331 001 84 ×10 4 C⋅mol −1. The Faraday constant can be thought of as the conversion factor between the mole (used in chemistry) and the coulomb (used in physics and in practical electrical measurements), and is therefore of particular use in electrochemistry. Since the 2019 redefinition of SI base units, the Faraday constant has an exactly defined value, the product of the elementary charge ( e, in coulombs) and the Avogadro constant ( N A, in reciprocal moles):į = e × N A = 1.602 176 634 ×10 −19 C × 6.022 140 76 ×10 23 mol −1 = 9.648 533 212 331 001 84 ×10 4 C⋅mol −1. It is named after the English scientist Michael Faraday. Solution: The charge of one electron is 1.6025×10-19C. 
In physical chemistry, the Faraday constant (symbol F, sometimes stylized as ℱ) is a physical constant defined as the quotient of the total electric charge ( q) by the amount ( n) of elementary charge carriers in any given sample of matter: F= q/ n it is expressed in units of coulombs per mole (C/mol).Īs such, it represents the " molar elementary charge", i.e., the electric charge of one mole of elementary carriers (e.g., protons). Remember that this charge is always in units of Coulomb (C) and the mass we take here is in Kg (kilogram).The electric charge of one mole of elementary charges A neutron is a neutral atom with 0 charge.  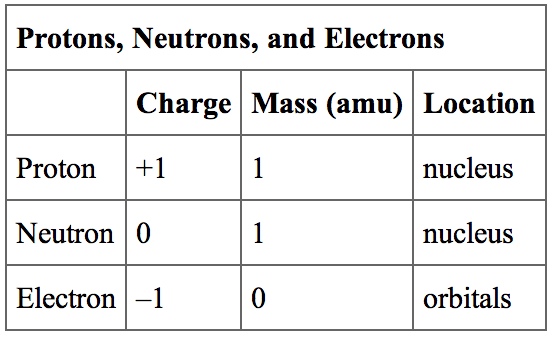
Hint: We know that 1 mole of any substance will have $6.02 \times $C.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
